
Erik Musiek, MD, PhD
Charlotte & Paul Hagemann Professor, WashU Neurology
- Phone: 314-747-5412
- Email: musieke@nospam.wustl.edu
Aging, oxidative stress, and circadian clock genes in neurodegenerative diseases
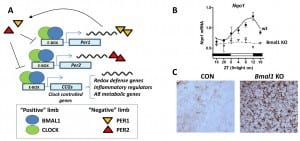
Aging and oxidative stress have been implicated in neurodegeneration, though the proximal mechanisms linking these factors are incompletely understood. Circadian clock proteins regulate many aspects of cellular biochemistry and have been implicated in control redox status and aging. Importantly, circadian clock gene expression and function decline with age and in neurodegenerative conditions. However, the role of clock genes in brain health and neurodegenerative disease pathogenesis is unknown. Our hypothesis is that the core circadian clock genes regulate critical aspects of brain redox homeostasis, bioenergetics, and metabolism, and that disruption of clock genes might facilitate neurodegeneration. We have observed that deletion of the master clock gene Bmal1 in mice, which completely abrogates circadian rhythmicity, induces age-dependent neuropathology characterized by neuronal oxidative damage, neuroinflammation, impaired antioxidant gene expression, and degeneration of synaptic terminals. Our lab is actively investigating the molecular mechanisms by which circadian dysfunction may contribute to brain aging, oxidative damage, and neurodegeneration.Circadian dysfunction is a common symptom of Alzheimer’s Disease (AD), and epidemiologic and genetic studies suggest a possible role for clock gene dysregulation in AD pathogenesis. Using mouse models of AD, we are examining how perturbation of clock gene function impacts amyloid-β metabolism and other aspects of AD neuropathology. Ultimately, we hope to develop therapeutic strategies for AD which target circadian gene function.