Constructing and packaging viral vectors for experimental gene delivery
About
The Hope Center Viral Vectors Core provides lentiviral and AAV vectors and other services to Washington University investigators on a fee-for-service basis. Investigators from nonprofit/academic institutions outside Washington University may use Core Services as well. The Core has special expertise in the use of viral vectors for neuroscience applications. The Core also supervises shared molecular and cell culture equipment in the Hope Center.
The goals of the Core are to provide the highest quality viral vectors available, to serve as a repository for the viral resources and expertise available at Washington University and to develop new vectors and reporters for neuroscience research.
Scroll down to learn about additional services and vectors available, as well as suggested language for acknowledging core use.
Services
The Core assists investigators with the design and construction of lentiviral and AAV vectors, and with small or large scale viral packaging. We provide viruses expressing marker proteins for use in testing viral expression in different cell types and organs. Basic reporter vectors are available. Investigators should obtain Recombinant DNA approval from Environmental Health and Safety prior to using these vectors in their laboratories. Labs that have not yet obtained approval can perform some experiments in the Core facility with training and supervision by Core staff. Vectors are not available for for-profit or human diagnostic applications.
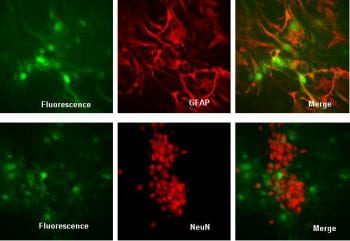
Lentiviral vectors are now widely used for modeling brain diseases, for genetic pharmacology and for delivery of potential therapies into animal models. The Hope Center Viral Vectors Core uses third generation HIV-1 based lentiviral vectors. These vectors have been used successfully in rodent models of lysosomal storage diseases (Hofling et al., 2004) in cultured neurons (Lee et al., 2004) and in PNS cell types such as Schwann cells and dorsal root ganglion cultures (Abdulkadir et al., 2002; Modur et al., 2002; Araki et al., 2004; Schalch et al., 2004; Le et al., 2005; Bermingham et al., 2006). Third generation lentiviral vectors offer several advantages: they readily transduce CNS neurons and glia, lymphocytes, endothelial cells and stem cells; they have very low toxicity, induce a minimal immune response, and are expressed for prolonged periods (6 months or longer, Blomer et al., 1997). Vectors used currently have a moderate cloning capacity (up to 8 kb) and can be generated with dual promoters or with internal ribosome entry sites (IRES) to enable expression of two transgenes from the same mRNA (e.g., the FCIV vector used here, see Araki et al., 2004).
Lentiviral vectors have also been very useful in the delivery of silencing RNAs. The core can help investigators design and construct new short hairpin RNAs (shRNA) for delivery in lentiviral vectors, or can package lentiviral shRNA vectors obtained from other sources, such as Sigma’s Mission shRNA.
Adeno-associated virus is nonpathogenic in humans. Recombinant AAV vectors typically do not elicit a cellular immune response. AAV vectors are able to spread widely in vivo after injection and maintain long-term expression of transgenes. Various serotypes of AAV vectors allow tissue specific delivery of genes of interest (Cearley et al., 2005; Harding et al., 2006). AAV vectors have become increasingly important in neuroscience research and clinical applications.
AAV vector serotypes 1, 2, 5, 6, 7, 8 and 9 are available in the Core. For AAV vector serotypes 7, 8 and 9, users must obtain an MTA from University of Pennsylvania before use. The following promoters are available for each serotype of AAV vectors: CAB (chicken β actin promoter with CMV enhancer), SYN (synapsin), and GFAP (glial fibrillary acidic protein). We will work with users to develop custom promoter constructs if needed.
Suitable for in vitro applications
Vectors available for purchase
The Hope Center Viral Vectors Core is pleased to announce availability of AAV vectors for Optogenetics. These vectors were chosen for production by the Viral Vectors Core based on responses to a survey last summer, and now are available for purchase for $200/aliquot (or $128/aliquot for Hope Center faculty members). The titer for each is at least 1 x 10^13 vg/ml.
- AAV5-EF1a-DIO-hChR2(H134R)-EYFP**
- AAV5-EF1a-DIO-hChR2(H134R)-mCherry**
- AAV5-EF1a-DIO-eArch3.0-EYFP**
- AAV5-FLEX-ArchT-tdTomato
Viruses can be used for in vitro Optogenetics (or in vivo applications in your lab). The Hope Center is developing in vivo Optogenetics as a core service – stay tuned!
MTAs and Acknowledgements
Deisseroth vectors
Vectors marked ** were created by Karl Deisseroth and we are pleased to stock these constructs. First-time users must obtain an MTA directly from Stanford. Click here for instructions on how to obtain an MTA.
tdTomato vector
The tdTomato vector was created by Edward Boyden (MIT), and generated by the Hope Center Viral Vectors Core using a plasmid from Addgene. An MTA is not required for the tdTomato vector. When publishing results based on use of this vector, please acknowledge the Principal Investigator, cite the article in which the plasmids were described, and include Addgene in the Materials and Methods of your future publications.
Suggested language for Materials & Methods:
pAAV-FLEX-ArchT-tdTomato was a gift from Edward Boyden (Addgene plasmid # 28305)
Suggested language for References:
A high-light sensitivity optical neural silencer: development and application to optogenetic control of non-human primate cortex. Han X, Chow BY, Zhou H, Klapoetke NC, Chuong A, Rajimehr R, Yang A, Baratta MV, Winkle J, Desimone R, Boyden ES. Front Syst Neurosci. 2011;5:18. Epub 2011 Apr 13. 10.3389/fnsys.2011.00018 PubMed 21811444
Contact information and feedback
We always strive to improve services and resources offered by the Hope Center Cores. To provide feedback, see contact information below.
Core Director: Mingjie Li, PhD
Core Facility Phone: 314-747-1123
Ideas for additional vectors?
If you’d like to suggest additional vectors for bulk production, please contact Mingjie Li.
Initiating use
Click below to learn more about available vectors, or for a new virus request form. For questions, contact Mingjie Li.
NOTE: Beginning July 2022, users must provide a cost center number when requesting a new virus.
User fees
Charges for Hope Center investigators are subsidized by the Hope Center. This subsidy will apply only if the billing PI is a Hope Center faculty member.
Acknowledging the Core
Please acknowledge the Core in manuscripts as well as posters and talks. Suggested language: “This work was supported by the Hope Center Viral Vectors Core at Washington University School of Medicine.”
ICTS Funding Option
Hope Center Core users may be eligible to apply for a Just-in-Time (JIT) award from the Washington University Institute of Clinical and Translational Sciences (ICTS). Faculty members without established funding for their projects are encouraged to apply for JIT funding through the ICTS. Visit the ICTS JIT webpage for additional details.